HairCell: New Website
(Updated 1/5/18 with Teslabrush Photos)
The electrifying startup, HairCell, has just launched its new website. HairCell is part of the Leonhardt Ventures portfolio, a venture creation lab that focuses on regenerative medtech and regenerative economy startups. The core technology of HairCell is a bioelectric stimulator device that controls expressions of specific proteins in sequence for regenerative purposes. In other words, it mimics the natural bioelectric signals that your body sends throughout itself to repair, and build, tissues and organs. The HairCell signals are sent out through an electrode equipped helmet.
Combining Approaches For Hair Growth
HairCell’s new website comes with some interesting new information. The company has developed three clinical trial protocols involving the HairCell stimulation technology.
- The first protocol includes using the HairCell stimulation technology in clinic and an electrostatic device for at-home use called the Teslabrush.
- The second protocol involves the two previously mentioned treatments plus amniotic fluid and/or PRP delivered via the Dermapen microneedling device in clinic.
- The third protocol includes everything from the second protocol and adds HC-15 fifteen component stem cell + growth factors + exosomes + micro RNAs + selected alkaloids + nutrient hydrogel + hair scalp/ECM matrix mixed hair regeneration composition.
This is a highly intriguing therapeutic combination. The amniotic fluid itself contains mesenchymal stem cells and growth factors. These would act as a “fertilizer” to go along with the Dermapen microneedle which activates the skin’s wound-healing mechanisms. Previous research has shown that wounding plays an enigmatic, yet positive, role in hair follicle formation. This Dermapen/fluid combination is added to the base therapy of bioelectric stimulation which is intended to stimulate the release of native proteins in the body for organ and tissue generation. The HairCell protocols comprise a thoughtful array of therapeutic techniques to stimulate hair growth. It all really comes down to a specific point of emphasis: Will this bioelectric stimulation device cause hair follicles to sprout, or rejuvenate vellus hairs back to terminal hairs?
HairCell’s Origin In Human Trials
One question that is always important to ask when being presented with a new hair regeneration therapy: where did this treatment originate from?
i.e. Why is it here? The FAQ page on the HairCell website gives some background as to where the initiative to create HairCell came from. Here is a quote describing a clinical trial led by the Leonhardt team which opened their eyes to the potential of hair regeneration:
“In 2012, 2014 and again in 2015 the Leonhardt led team initiated clinical studies in Czech Republic, Switzerland, Germany and Mexico for limb salvage and ulcer wound healing to stave off imminent amputation. In the Czech Republic study we were able to save 12 of 16 legs treated just one time with a stem cell composition. In the study in Switzerland and Germany we worked with a team in Denmark (Wetling) to apply non-invasive bioelectric stimulation to 47 patients with non-healing leg ulcers. The team achieved 95% healing of all ulcers in all patients at 8 weeks. In Mexico we were able to save 7 of 7 legs treated with just two bioelectric signals for only 2 hours 2X a week for 8 weeks. We observed in these leg studies that when the wounds healed and the blood flow improved, that their was also very healthy hair growth on the treated legs. This launched the start of HairCell in 2016.”
Before and after photo of a leg treated with bioelectric stimulation.
This would make HairCell the second hair-growth treatment we’ve heard of that was discovered during diabetic leg ulcer trials. 5 bonus points if you can name the first one.
Clinical Ready To Go
The good news is the HairCell team is currently initiating clinical trials in North and South America to answer all of our questions. At least one of these trials is scheduled to read out data around the end of 2nd quarter 2018. Even better news is the bioelectric stimulator device has a previous FDA 510(K) approval. This means if the HairCell therapy proves to be safe and effective in its clinical trials it could potentially enter the market shortly following the trials’ completion.
Update: Teslabrush
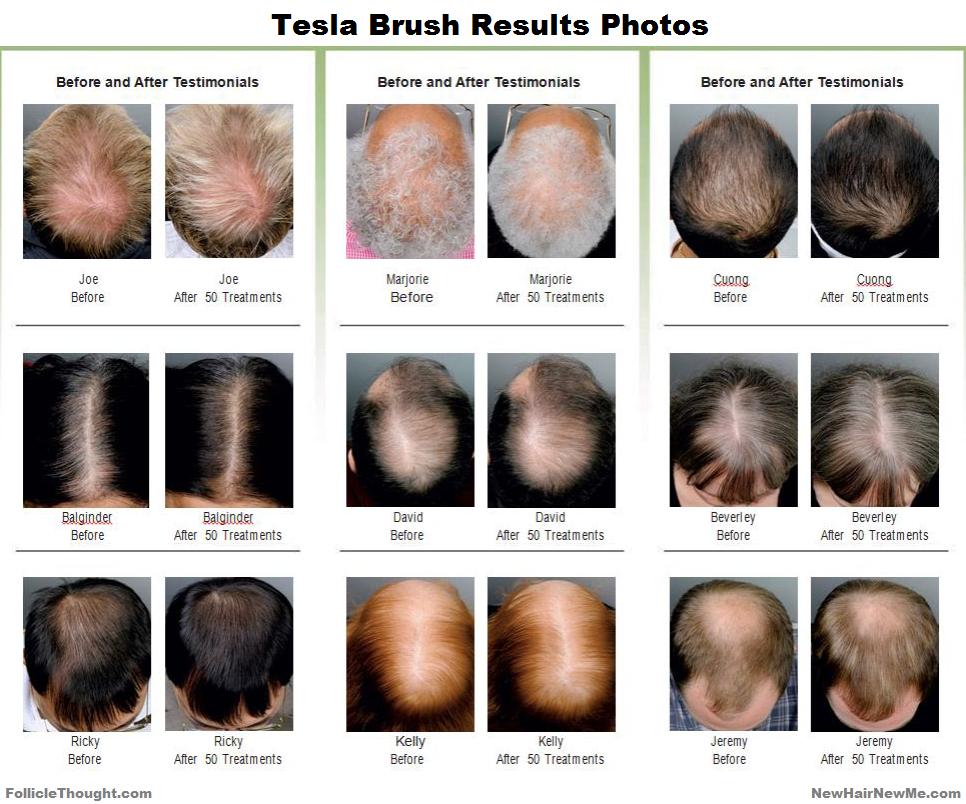
In response to commenter “Michael” (thanks again) I reached out to Bernhard Rudert for more information and asked him to share photo results of the Tesla Brush. Bernhard is an electrical engineer and the inventor of the Teslabrush. First, I will copy and paste Bernhard’s reply from the comments thread and following that I will share some before and after photos of the initial Teslabrush model.
“Hi Michael,
I fully understand your sentiment. When I was asked by a very successful wellness clinic to design a device for them I was hesitant because it did not look like a worthy project for a professional engineer. Soon I changed my mind when I saw the results from an experimental device. It was not an easy task to develop a device with the same characteristics while meeting mandatory regulatory requirements.
People loved the device. However, I still had a problem in that we engineers work with concrete facts. I had to learn to get used to the thinking of pharmaceutical companies where in many cases only a small percentage of patients can be helped with a given drug. However, the percentage was significant enough to make me go ahead with confidence. One thing that stood out was the fact the majority of users benefited from stopping or reducing hair loss and at least quite a good number got some hair back.
We still know very little about electrical stimulation regarding hair loss. I simply copied the stimuli of the experimental device realizing that there needs to be big investment to find an optimum. I use an electrical field to generate the pulses in comparison with devices which use direct electrical contact with the skin. The advantage is that the penetration into the skin is much more effective even in the presence of existing hair or dry skin.
There is still considerable research required as to why this device works. However, we see that with some users the skin takes on a faint pink color indicating that blood circulation is enhanced. Furthermore, after a while very tiny brown spots appear around dormant follicles indicating that excessive sebum is excreted from the follicle. Due to the electrical stimulation it can safely be assumed that on the cellular level more ATP is produced which makes more energy available for the process. I believe however, that the major benefit is derived from a neurological process which aims at restoring homeostasis.
Including the Teslabrush in the study is very sensible because it is very likely that it will enhance the other processes. I am very much in favor of a multi-pronged approach to hair loss. We don’t have a single pill that will solve the problem and from the immense pool of knowledge with the contributors this study could well lead to one of the most successful hair loss treatments available to date.
I have not put any photos on my website because they will be copied by those using scam methods. For this reason a summarized version of the original study has been presented. When trying to find a solution for hair loss your critical mind is a very useful tool.
I hope this answers some of your fully understandable skepticism.”
What I see here are some realistic, worthwhile results from a product intended to support hair growth. Obviously, ‘Ricky’ and ‘Cuong’ received advantageous results using the device. It seems to be an improvement from a hand-held LLT hair growth device. We could do ourselves a service by treating the Teslabrush as it is, a product intended to support hair growth and prevent loss. There are no inflated claims being made by the Teslabrush company. Because of the company’s credibility, Follicle Thought has agreed to affiliate with Teslabrush.
Before and after photo of a leg treated with bioelectric stimulation.
Posted in HairCell


great jop
HaHa,, lm going to be in the upcoming US trials this second quarter,,, the company told me they had initiated trials in Canada late this year and l’m assuming they were successful as the US trials are going ahead..Bite me Baldness!!
Best of luck Steve!
Iam very excited for you Steve. Keep us posted with your progress! Good luck. Denise
Steve, what is your Norwood type? Also, will this have to go through Phase 1, 2 and 3 clinical trials with the FDA? Are you in the first Phase?
Thanks much in advance.
OH? my Norwood scale makes telly Savalis look like a Chia pet… this phase trial is the final hurdle before approval later this year.. This is a cellular rebuild on a cue ball canvass.
They’ve identified the individual signalling frequencies for follicle formation. The principle is the same for every single organ.This North American trial will succeed.
Well, Steve is certainly confident. Let’s see how the trial goes.
Hi Steve,
I am sincerely grateful for your response.
It gives me hope because my Norwood type is like Danny DeVito (the actor). So I am at the bottom of the pile. LOL.
Anyway, you said “approval later this year”. Did you mean FDA approval in the USA? Thanks again and please don’t be a stranger. Your good actions will be good karma. May you get back your full head of hair and God Bless you!
Trevor
Interesting.
Good morning,
I am extremely pleased to hear this, hope it all goes well and they won’t need 100 clinical trials.
Steve, please keep us updated.
Best regards!
Sounds promising, but potentially pricey.
The tesla brush makes this treatment seem a little new agey and scammy.
Michael, I think Bernhard can do a better job of explaining it than I can. Once again, thanks for commenting and leading towards a solution for your concern. I’m going to be sharing the photos he sent me tomorrow. (delayed, now Friday)
Hi Michael,
I fully understand your sentiment. When I was asked by a very successful wellness clinic to design a device for them I was hesitant because it did not look like a worthy project for a professional engineer. Soon I changed my mind when I saw the results from an experimental device. It was not an easy task to develop a device with the same characteristics while meeting mandatory regulatory requirements.
People loved the device. However, I still had a problem in that we engineers work with concrete facts. I had to learn to get used to the thinking of pharmaceutical companies where in many cases only a small percentage of patients can be helped with a given drug. However, the percentage was significant enough to make me go ahead with confidence. One thing that stood out was the fact the majority of users benefited from stopping or reducing hair loss and at least quite a good number got some hair back.
We still know very little about electrical stimulation regarding hair loss. I simply copied the stimuli of the experimental device realizing that there needs to be big investment to find an optimum. I use an electrical field to generate the pulses in comparison with devices which use direct electrical contact with the skin. The advantage is that the penetration into the skin is much more effective even in the presence of existing hair or dry skin.
There is still considerable research required as to why this device works. However, we see that with some users the skin takes on a faint pink color indicating that blood circulation is enhanced. Furthermore, after a while very tiny brown spots appear around dormant follicles indicating that excessive sebum is excreted from the follicle. Due to the electrical stimulation it can safely be assumed that on the cellular level more ATP is produced which makes more energy available for the process. I believe however, that the major benefit is derived from a neurological process which aims at restoring homeostasis.
Including the Teslabrush in the study is very sensible because it is very likely that it will enhance the other processes. I am very much in favor of a multi-pronged approach to hair loss. We don’t have a single pill that will solve the problem and from the immense pool of knowledge with the contributors this study could well lead to one of the most successful hair loss treatments available to date.
I have not put any photos on my website because they will be copied by those using scam methods. For this reason a summarized version of the original study has been presented. When trying to find a solution for hair loss your critical mind is a very useful tool.
I hope this answers some of your fully understandable skepticism.
any new about brotzu? it’s 2018 already..
Cris, thanks for asking.
Fidia has been very quiet throughout the process. I would guess some news comes around April.
Not sure if it’s possible Admin but it’d be great to somehow track any users progress that bite on the Tesla brush. I’d be tempted but it’s just a bit dodgy to me with the current website, results, etc.
Sure, Yoda. I’d have to hear from users who try it and be in touch with them. But I would certainly house user progress reviews here so long as the conversation stays civil.
Hi Yoda,
You are doing well in carefully investigating hair loss products before you use them. There are many clever advertisers who sell stuff that simply does not work. I tried hard to do some marketing but I realize that I do not have the skills. I will have tom outsource this activity. I spent a lot of time and money on the product based on the good results I have seen and I firmly believe that the Teslabrush should be in the arsenal of any person fighting hair loss. This is not only because it stops hair loss in most cases but facilitates the effectiveness of other hair loss products. Even people with hair transplants can benefit.
To get a more comprehensive picture of the performance of the device we cannot go by the results from just a few users. We would need a big clinical study of several hundred people the cost of which would be prohibitive and the price of the product will go sky high. Also keep in mind that in the medical field not every remedy works every time and for everyone. It is the same with the Teslabrush some people get fantastic hair growth and others only stop loosing hair. For good results we also need some faith in the product. It is the body that heals itself and the remedy serves as a catalyst.
Hello
Just like to say thanks for all of the current and useful info on this site.
I’ve noticed on the Tesla Brush site you can actually buy the brush. However I thought it was still in testing, unless I’ve misread/understood?
Many thanks
Clive
Yes you can buy the Teslabrush from the website. For the Hair Cell clinical study we want to see how far it supports or facilitates the process.
Bernhard,
Appreciate your response, I will take my time and see if there’s any feedback via users who visit this site (or other sites). In the meantime, please advise if someone uses topical treatments (minoxidil, etc. ) would the Teslabrush still be used at night after up to two applications per day of topicals? I see the protocol is for 10 mins, is this everyday?
Hi Yoda,
With laser devices it is important to have high power output in order to get results. This is difficult with a laser brush and a laser hood would be better. They call it laser light which has a very narrow wavelength spectrum approaching monochrome light. To my knowledge the cells in the human body do not distinguish between laser and normal LEDs. The LEDs used for commercial devices are standard devices not made specifically for hair loss treatment. Their wavelength is shorter than the preferred 660-700 nano-meter range. I have no comparative data but the Teslabrush is probably superior to commercial laser treatment requiring less treatment time.
You can combine the Teslabrush treatment with minoxidil. I suggest you apply the minoxidil after the massage with the Teslabrush. In some cases it might even be possible to get better results by using the combination than just using one method at the time only. Never stop using the Teslabrush when introducing other treatments because the Teslabrush enhances other treatments.
Thanks Bernhard, Ok like a laser device it seems like you recommend on a clean scalp (pre-minoxidil). I shower in the am so I’d have to use then as opposed to the eveing before bed like you’d indicated. Can you please answer the question, does this need to be used 10 mins a day…Everyday?
Hi Yoda,
It is the total treatment time that counts. Good results are also achieved by using the Teslabrush for 20 minutes every second day. You can do the treatment any time of the day but doing it before bedtime provides relaxation which helps with sleeping. Doing it daily at a certain time for a certain period creates a habit which makes it easier to be consistent. There is no permanent cure for hair loss and if you want to nuture what you have and even grow additional hair the Teslabrush will be a very economical long-time companion.
Hi Clive,
The Tesla brush has been added to the “HairCell Protocol” which is a multi-faceted approach consisting of bioelectric stimulation via electrodes(the core HairCell technology), microneedling + amniotic fluid, and the Tesla brush. This entire protocol is being put through a clinical trial to see how it all works in conjunction and also how certain parts of it work themselves. For more info visit the new HairCell website.
Thanks for the reply.
Ah I see, I’ll take a closer look at the new site.
Wonder if the Teslabrush works on its own or better than the laser brushes? But I know there is no silver bullet yet!
Thanks
Clive
Hi Clive,
It is difficult to get the required output power from a laser brush unless one spends a lot of time massaging the head in order to compensate for lower power. A laser hood would be a better solution but it still requires long treatment times and the wavelengths generally are not optimum. Also darker skin does not allow the light to penetrate deep enough to get results.
The Teslabrush can be used on its own. When used with some herbal remedies or supplements these additional products work more effectively. The Teslabrush has been designed for long term use because I believe that everybody suffering from hair loss will want to use it for many years even in addition to other treatments.
My only comment back Berhard is that I have one of the original Hairmax laser brushes and using it even for approximately 5 mins, 3 days a week is a pain in the you know what. I can’t imagine having to use a brush for 10 mins a day or 20 mins every other day. I guess if it was proven to regrow a ton of hair then that might blunt the inconvenience. I’ll wait to see more data and testimonials from actual users before I bite. The only reason I still use the Hairmax is that it’s a sunk cost at this point, bought it approximately 15 years ago, it has limited effect if any at all.
All the answers are in this Follicle thought story with before and after shots.. https://folliclethought.com/haircell-new-website/ The video on the Tesla brush site said everyday for 10 mins..
The before and after shots are not impressive as far as regrowth is concerned but this brush may be very effective as a preventative, as it excites cellular activity and blood flow very well.. lt is also a wound healer and skin regeneration device..
On the Haircell websites (old and new) there is mention of a large array of growth factors (VEGF, IGF-1 etc etc) that are pumped into the scalp at the same time as bioelectric stimulation is carried out. As far as I can tell these are not being used in the trial, does anyone know if this is accurate and if so why they have decided not to use these substances?
Hi Matthew,
I’m not sure why the switch has been made, perhaps for regulatory purposes, but it seems like delivery via microneedling has taken its place.
Hi thanks for getting back to me, are you suggesting that they’re still using the same growth factors but delivering them via a different method? I couldn’t see any mention of them but maybe I missed something…
The protocol has changed a lot and I’m not sure, so I don’t want to speak for them. But I believe the 3rd protocol on the new website mentions the original treatment plan.
Has anyone used the Tesla Brush and if so do they have any comments on feedback on the results? The only information I can find when I search dates back to 2009…
Much appreciated.
Ok I was pretty skeptical because not had I only never heard of such a phenomenon as using electrostatic impulses to help grow hair back but I’m sorry to say (I have no intention of sounding rude here) but the name “Tesla Brush” kind of made me cringe when I first read it. I have heard of the experiments Tesla did with electricity but it just seemed like a marketing tactic to have his name in the product. I have done a little bit of research since then and it does look like a study was done with hair growth and electrical impulses.
https://www.ncbi.nlm.nih.gov/m/pubmed/2397975/
What compelled me to look into this was Bernard’s honesty in saying that this is no cure and may not work for everyone. In life you have to take chances if you want to move ahead so I have to say that I have moved from skepticism to seriously considering this product.
Hi Michael,
I appreciate your comment. Electrical and even more so electrostatic stimulation for many kinds of ailments is a science which is still at its beginning stage. I intend to do much more research in this field.
Michael, if you’d be so kind to let the readers of this blog know:
1. If you move forward and purchase a Tesla Brush.
2. Your thoughts while using.
3.If using 10 mins a day or 20 mins every other day is feasible and you keep up with it regularly.
4. Results, results, results!!
It would be greatly appreciated as there is little to none unfiltered feedback from actual users to be found online.
Sure. I’m thinking of using it in addition with either RU58841 or Brotzu when it comes out. If I choose the latter – I still might use the tesla brush in addition to my current regiment which is just Nizoral and Segal’s and I might start derma rolling once a week.
Yoda advises not to wait for future treatments like the so called Brotzu…they come and go and may or may not be effective. I recommend hoping on the Big 3, if you’re worried about sides that’s a personal decision. I’ve been at this game for over 30 years and sides are way overblown on forms. As far as RU, I would never advocate rubbing unregulated Chinese chemicals on one’s noggin, however after much consternation Yoda has added this to my regime (30% pm and 15% am minox, Dut and Niz) due to lack of regrowth. To early to tell but I might be seeing some benefit…could also be wishful thinking though.
I’ve read derma stamping is better, as it avoids the rolling of hair around the roller..
I use the Derminator, still hurts like hell but gets the job done quickly. Recently changed my routine. Derminator 1.5 mm behind hairline and 2.25 mm on hairless temples every two weeks. Use a 0.50 mm roller every other day for absorption. We’ll see how this goes.
l know the derma stamping and rolling keeps the skin in a constant state of healing and increases circulation. What’s missing is the stimuli to cause follicular cell formation instead of skin cell reformation in this healing state. Stamping alone cannot be responsible for new follicle formation, unless an agent is applied in the healing phase to trigger the process of new follicle formation. That agent turns out to be AZT (the AIDS drug) and one company reviewed here is on the verge of releasing a system involving initial skin cell disruption (injury) followed by the application of an agent which will trigger the new hair follicle growth.
Has anyone began using the Tesla Brush?
Would like to hear feedback from anyone who has started using the device.
I just got mine in the mail and did my first session this evening. I will let you all know how it goes.
Awesome, thank you!
Hi Andrew, any update on your progress?
I got ordered one too. Will report back 🙂
Any development? Have they presented any data or indicated an estimated market date?
I believe they working towards initiating a clinical trial outside US. I will share an update when unavailable.
I’d like to invite anyone who has been using the Tesla Brush to please share some feedback, I have actually not received one single comment from any users who have began using the product. I’m interested to hear how it’s going. Thanks!
I’m very interested hearing some more reviews from people too. The lack of feedback is the only thing stopping me from trying it myself. So far I’ve only heard from one person who did, who mainly commented on its use as a massage tool, using the glass diodes for scalp kneading. He did not mention anything about regrowth, but did say his scalp elasticity improved and that it was well worth the money.
I purchased a unit in early June, so I’ve been using it 15 minutes per day for about 10 weeks. Not noticing any new growth yet, but after a little increase in shedding the first month, I would say further hair loss has significantly slowed down. I’m somewhere between Norwood 3 and 4, and now finding only a few hairs in the sink after combing in the morning post-showering. I’ve not used finasteride in a number of years but may go back on it to see if I can get some regrowth.
Thanks for the review Steve! Sounds like it has been of some value to you.
any further reviews or updates on the effectiveness of the tesla brush
How is the trial going Steve? it’s been six months… any report?
Hi. Been at it pretty much every night. Can’t say there has been any notable new growth, but things have at least been stable with minimal loss. Very few hairs fall out after showering and brushing now. As I have not added any new products, it would seem that there may be some effect here. I would say that I’m satisfied and hope this stays effective until something better comes along. If the Trinov reviews are positive over the coming months, that might be the next thing to try (would be nice if they get an American or Canadian partner to produce the product over here more economically for us though)..
Hello, is it possible to purchase the teslabrush anywhere ?
Jake, I don’t think the Teslabrush is for sale anymore.