RiverTown Therapeutics Inc. Clinical Update
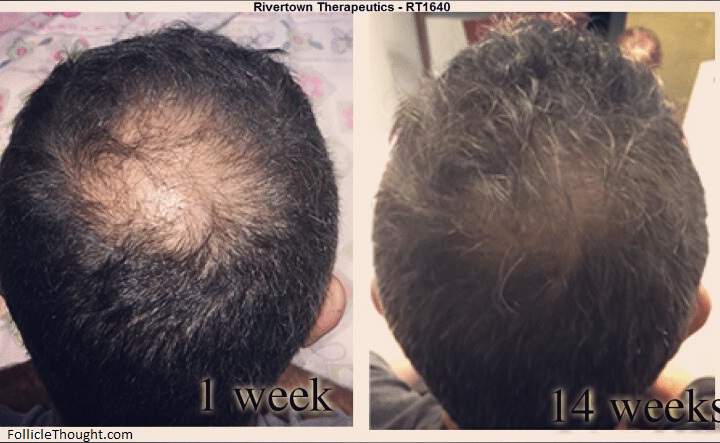
I’ve had many readers inquire lately about the current progress of RiverTown Therapeutics Inc.’s RT1640. RTI’s therapy for hair regeneration, RT1640, is a topical medication composed of minoxidil, cyclosporine A, and a novel molecule called RT175. The company debuted some impressive hair growth photos on this site last year. The question that’s been on a lot of people’s minds since then – “how’s things coming along at RiverTown?” I recently caught up with David Weinstein MD PhD, the CSO/CMO of RiverTown Therapeutics Inc. to give you all a clearer picture of what they’ve been working on and what the future may look like for this company of interest.
Q&A with David Weinstein MD PhD
FT: When/where is the next clinical trial for RT1640 taking place?
DW: We are currently negotiating a Phase 2 trial in Australia. The Australian government has made a concerted and focused effort to enhance the biotechnology effort in their nation. To that end, they have made it both easy and very cost-effective to carryout trials there by granting very liberal tax incentives and R&D rebates to small companies. We will let you how these negotiations progress.
DW: Alopecia areata is an autoimmune disease. Like most autoimmune diseases, the underlying etiology is unclear. We know that this is a systemic disease, in which T cells recognize elements of the hair follicle as being foreign, and attack them, resulting in lots of inflammation and hair loss. We also know that people with one autoimmune disease are more prone than others to develop additional autoimmune disorders, and that this is particularly true when the first disease is very active and there is a lot of inflammation. While I believe that RT1640 will induce additional hair growth, I am concerned that such growth will provide “fuel” for the underlying autoimmunity, and “rev”up the system, resulting in more inflammation. As a physician, my first priority is to do no harm. I am concerned that growing hair in patients with alopecia areata will do harm as I described. Therefore, we have no plans to explore this indication.
Having said this, we have some preliminary evidence that RT1640 treatment does restore hair growth in the setting of taxane-induced “permanent alopecia.” These are people that have been treated with taxane drugs to treat cancers- usually breast cancers- and have suffered what has been thought to be an irreversible alopecia. More on this as we learn more.
FT: Will RT1604 be effective on areas of the scalp other than the crown, such as the frontal scalp/hairline?
DW: The distribution of androgenic alopecia is a function of two factors: the concentration of androgen receptors (ARs) and the concentration of the testosterone to DHT conversion enzyme 5AR. There is good evidence that the areas with the highest degree of androgenic alopecia are the areas with the highest concentrations of both ARs and 5AR. Unfortunately, these tend to be at the temple and the crown. We have not studied the effects of RT1640 in enough people to say for sure, however, our anecdotal evidence suggests that our therapy will regrow hair at the temple, and on the crown for certain.
DW: Recent work from Dr. Lu Le the University of Texas has shown that the transcription factor krox20 expressed by cells in the hair follicle is essential for the growth of new hair. Throughout my career I have studied another transcription factor, called Oct6. In every tissue we have ever found Oct6 expression, it is closely associated with the expression of krox20. In fact, krox20 expression is dependent upon cells first expressing Oct6. Our work shows that treatment with RT1640 strongly induces Oct 6 expression. This finding supports our hypothesis that RT1640 treatment induces molecular changes in the hair follicle that drive new hair growth.
Thank you David, for taking the time.



Hello,
Thank you for the good news, yet, maybe it’s my eyes but on the second picture I see a little ‘wash’ on the crown area, like the picture has been slightly blurred right at this point. Of course, the result is amazing and gives great hope. Thank you.
No wash on the photo, just put through a lighting filter to brighten it up a bit. The original is on RT’s website.
Alright good 🙂 thank you for your amazing site and hope you give to all of us Admin, we’ll win the fight against hair loss ..
Look like different heads to me…
It’s not different heads lol
2021 or 2022, wow that’s super fantastic news!
Glad you’re happy about that Keith
2021 or 2022 as everyone says every effective treatment just 5 yrs away. …can’t bear more.
I hear you Vivek, like wise here, we will vanquish, we will..
ahah!
hope is drying up for me. If tsuji fails im going for smp!
the only hope for a real cure is tsuji/riken /kyocera
forget this.
admin can you take an interview with dr. tsuji?
I guess I would have to say I am “amused” at the perspective some people have about prospective treatments. This therapy is beyond what any other topical has produced thus far and is likely on par with the best injectable therapies. Tsuji will be great when it perfects the art of cloning a hair follicle outside of the body. I’m hopeful, though how long could that take?
riken/kyocera aim to 2020 in market (japan first)
Yes, that is their goal. Wish them all the best.
Do you know what percent of the solution will be Minoxidil?
Nope. 5% seems likely.
I must agree with admin and say Im intrigued with the attitude from people in hair loss forums, here and elsewhere. I’m having a hard time seeing the point of visiting forums to just kick down on news and potential treatments with the argument in lacking faith, without any references to actual science and data. We all want a perfect solution, some easy sci-fi stuff where this just goes away. But if you are so disillusioned and pessemistic that you just have to kick down on valid results and potential candidates that can help, why not just shave your head and get on with your life? Sure, hair cloning and 3D printing and complete recoveries sounds awesome, and I find them scientifically significant and interesting even in science own right, but that may be 20-30 years away before it is perfected and available to the average citizen. So why not be a bit realistic and keep some hope up? Nothing is going to change because of this negative attitude. Shape up, shave or get a transplant if you are in a hurry, no miracle cure will show up on your doorstep because you are sulking.
Keep up the good work admin
??
Hey joeseph, looking good with rivertown also a very interesting news on your twitter account. You consider that another wild card?Do you know if they would have to go threw clinical trials or go to market considering it was FDA approved years ago? Exciting times coming.
Thanks for the interest Chris. Not a wild card, but could go quickly through trials if funded.
great work with the last two twitter tweets.
Any update on this?
Not at the moment.
Certainly promising. You have to ask yourself why they don’t use a flash for both photos though…
Been researching how I can get my hands on this stuff. Even DM’d Rivertown. Wish the FDA would be more liberal to allow private citizens to volunteer after phase 2 and sign waivers in order to contribute study data* and move the thing forward.
Wow! The photos are truly remarkable! They give me great hope. Are there going to be clinical trials in the US? I’m a female who has suffered from hair loss for over a decade and I would love to be a guinea pig! As a woman, it’s traumatic loosing your hair!
I understand Palatina, I wish you all the best in your recovery for hair. I was just speaking to RT the other day and they are aiming for trials in the US instead of originally planned Australia. However, it all depends on them getting the funding for the trial. Keep your fingers crossed 🙂
Reader’s Opinion:
Is there any other topical that is close to or better than RT1640?
Does anyone have an opinion on RCH-01 being able to produce these results? You think it’s possible?
Are there anymore updates? Is 2022 still the projected release date?
They’re still working really hard for financing! 2022 is still possible if they could get funded asap. They need to do a phase 2 and then have a larger partner take it through to phase 3.