Updates
Cures For Hair Loss 2023 - News Feed
A very good idea is to bookmark this article page in your browser or mobile browser for easy access on the latest hair loss cure and hair growth treatment news. This will allow you to check it easily for updates. If you are looking for a corresponding article from a past update, simply use the “Search” function on this site to locate the article. As always, thanks for reading and please share this information with all you feel would benefit from it!
OliX Pharma Announces New Data at the World Congress for Hair Research
OliX Pharmaceuticals, a Korean biopharmaceutical company that focuses on developing RNAi therapies, has recently announced some results from its hair loss indication OLX104C (an asymmetric siRNA targeting androgen receptor therapy) at the 13th World Congress for Hair Research (WCHR 2024). A collaborator working with OliX on research on OLX104C, Dr Jonghyun Won, presented the data on the hair loss inhibition effect of the drug and the long-term impact of the drug in dermal papilla cells, human hair follicles, and mouse models.
While no results have been discussed in any press releases, they have been described as "promising", so we hope to get some published news soon.
The presentation at the WCHR was titled "Efficacy of Asymmetric siRNA Targeting Androgen Receptor for the Treatment of Androgenetic Alopecia".
Stemson Therapeutics and Aderans Announce Exclusive Licensing Agreement
Stemson Therapeutics and Aderans, the parent company of Bosley and HAIRCLUB, announced an exclusive global licensing agreement to develop and commercialize hair regeneration therapies on March 8, 2024. Stemson will leverage Aderans' Phase 2 clinical-stage cell therapy technology to enhance its product pipeline for hair loss. Aderans' platform uses patient-derived cells to rejuvenate hair follicles in individuals with androgenetic alopecia. Stemson will integrate Aderan's technology with its iPSC-derived cell therapy to create advanced solutions for various hair loss conditions.
Dr Bloxham – Verteporfin 5 Month Update
The wait is over for anyone interested in verteporfin. Dr. Bloxham has released a YouTube video update for his verteporfin experiment, which you can find here. We will also be updating the main article, so stay tuned.
Hope Medicine’s HMI-115 Positive Phase 1b Outcome
Positive news for those following Hope Medicine's HMI-115. The company has successfully completed a Phase 1b study in Australia, conducted in 12 male and 4 female patients with alopecia areata. The results showed positive efficacy, with HMI-115 being safe and well-tolerated. In male patients, the mean non-vellus target area hair count (TAHC) increased by 14 hairs/cm2 by the end of the trial, which was statistically significant compared to the baseline measurements. However, no results have been released yet for the female participants. These results have led to the start of recruitment for the Phase 2 study, which will be conducted in China with 180 participants.
Epibiotech Approved to Start Phase 1/2a Trials
As of December 21st, 2023, Epibiotech has been approved by the Ministry of Food and Drug Safety to start Phase 1/2a clinical trials for its EPI-001 autologous dermal papilla cell treatment to treat androgenetic alopecia. Safety will be measured in Phase 1 to set the maximum tolerated dose (MTD), and then the effectiveness of the treatment will be evaluated in the Phase 2a trial. Epibiotech plans to administer the treatment to the first patient in the first half of 2024, with completion expected in the second half of 2025.
Yuva Biosciences Enters into a Collaboration with Elevai Labs
On November 29th, 2023, ELEVAI Labs, a medical aesthetic company, announced that they had signed a licensing agreement with Yuva Biosciences, Inc. This will combine ELEVAI's human umbilical mesenchymal stem cell-derived exosome technology with Yuva's mitochondrial research in an effort to develop pharmaceuticals and cosmetics targeted toward aging hair and skin. The collaboration might allow for the development of new products to address cosmetic skin and hair concerns associated with signs of aging.
Kintor Pyrilutamide KX-826 Phase 3 Results Shared
The world's current furthest developed drug for androgenic alopecia prior to approval, Kintor's pyrilutamide aka KX-826, has completed its phase 3 trial for men in China. A non-detailed explanation of the trial's data was sent out by Kintor Pharmaceutical on November 27, 2023. In short, pyrilutamide showed overall safety in the trial with no serious adverse events reported, the drug also showed an increase in target area hair count (TAHC) in the treatment group compared to their baseline numbers, and it also showed improvement in TAHC over placebo during all treatment visits of the trial, though they were not statistically significant. Therefore, overall KX-826 did not show a statistically significant improvement in TAHC compared to placebo. The future of pyrilutamide for AGA is now uncertain.
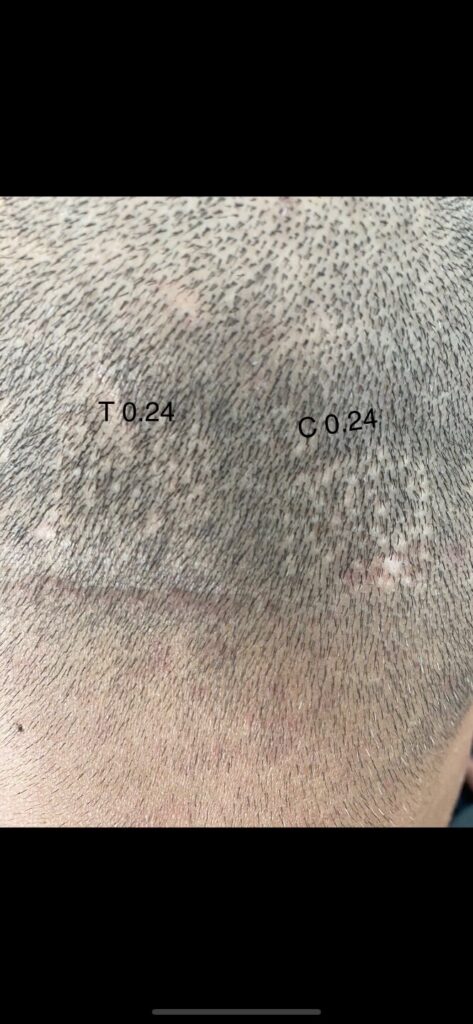
Dr. Barghouthi Verteporfin Study Updated
The groundbreaking verteporfin study done by Dr. Barghouthi of the Vertex Hair Clinic in Jordan has recently been updated with new photos. For the newcomers, the study involved injecting the drug verteporfin into FUE donor area extraction sites, i.e. wounded scalp skin. Based on preclinical research at Stanford, it is believed that verteporfin has the potential to decrease or eliminate scarring, and/or regenerate hair follicles. Below is the least impressive of the recent photos shared by Dr. Barghouthi. Check out the original documented post on Follicle Thought to see the higher concentration of 0.4mg injections ~1.5 years post surgery.
Veradermics Doses First Patient With New Oral Hair Loss Drug
A fascinating new dermatology startup, Veradermics, announced today the dosing of its first patient in a phase 2 clinical trial for pattern hair loss. What makes this news especially exciting is that Veradermics' drug, for now referred to as VDPHL, is an oral medication. Further, it's a non-hormonal drug, which means it does not act on androgen hormones, such as DHT, like the drugs finasteride or dutasteride do. This means that users will likely not have to worry about sexual-related side effects. While information about VDPHL is limited at this time, Veradermics describes it as a "proprietary, non-hormonal oral treatment being developed for pattern hair loss." Stay tuned for further information about this drug program here on Follicle Thought.
HMI-115 Starts Phase 2 Clinical Trial In China
As the title says, HMI-115 is now at the phase 2 clinical trial stage in China. The placebo-controlled trial will include a 6 month treatment period for its participants. For now, this news seems to be the capstone of a quite active and positive year for hair loss treatment companies. To read the full story, check our last article on Hope Medicine.